NON-DUALITY
“If all this damned quantum jumping were really here to stay, I should be sorry, I should be sorry I ever got involved with quantum theory.”
— Erwin Schrödinger
Many physicists in the late nineteenth century were frustrated until, finally, one of them broke rank: Max Planck, of Germany. In 1900, Planck took a bold conceptual leap and said that what the old theory needed was a quantum jump. (He borrowed the word quantum, meaning “amount,” from Latin.) What emitted the light from an incandescent body—burning wood, for example, or the Sun— were tiny jiggling charges, the electrons. These electrons absorb energy from a hot environment, such as a fireplace, and then emit it back as radiation. This part of the old physics was correct, but then classical physics predicts that the emitted radiation should be rich in ultraviolet, which is contradicted by our observations.
Planck declared (very bravely) that if the electrons are assumed to emit or absorb energy only in certain specific, discontinuously discrete amounts—which he called “quanta” of energy—the problem of the emission of varying degrees of ultraviolet could be solved.
To explore the meaning of the quantum of energy, consider an analogy.
— Erwin Schrödinger
Many physicists in the late nineteenth century were frustrated until, finally, one of them broke rank: Max Planck, of Germany. In 1900, Planck took a bold conceptual leap and said that what the old theory needed was a quantum jump. (He borrowed the word quantum, meaning “amount,” from Latin.) What emitted the light from an incandescent body—burning wood, for example, or the Sun— were tiny jiggling charges, the electrons. These electrons absorb energy from a hot environment, such as a fireplace, and then emit it back as radiation. This part of the old physics was correct, but then classical physics predicts that the emitted radiation should be rich in ultraviolet, which is contradicted by our observations.
Planck declared (very bravely) that if the electrons are assumed to emit or absorb energy only in certain specific, discontinuously discrete amounts—which he called “quanta” of energy—the problem of the emission of varying degrees of ultraviolet could be solved.
To explore the meaning of the quantum of energy, consider an analogy.
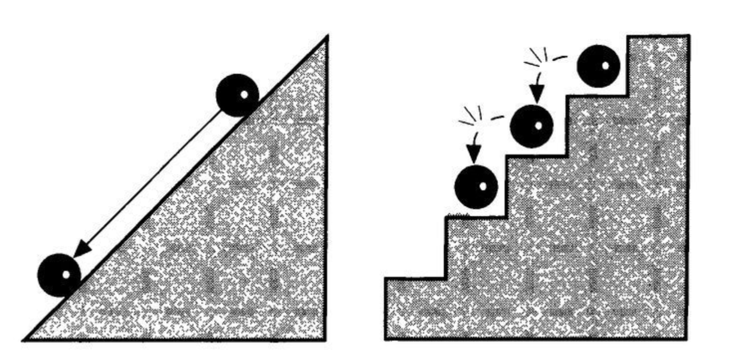
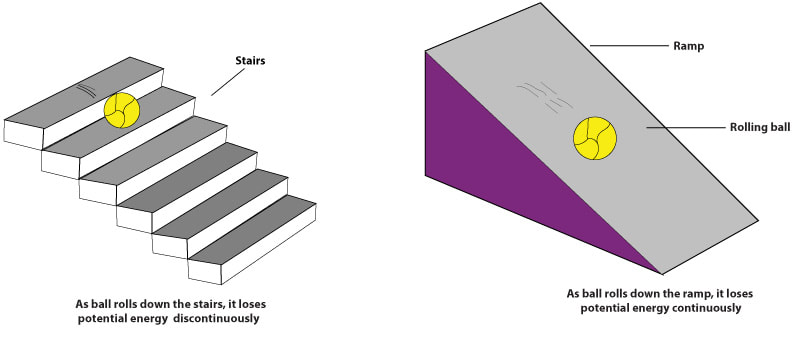
Compare the case of a ball on a staircase with one on a ramp (fig. 1). The ball on the ramp can assume any position, and its position can change by any amount. It is, therefore, a model of continuity and represents how we think in classical physics. In contrast, the ball on the staircase can sit only on this step or that; its position (and its energy, which is related to position) is “quantized.”
You may object. What happens when the ball falls from one step to another? Is it not taking on an intermediate position during the descent? This is where the strangeness of quantum theory enters: For a ball on a set of stairs, the answer is obviously yes, but for a quantum ball (an atom or an electron), Planck’s theory answers no. A quantum ball will never be found in any place intermediate between two steps; it is either on this one or on that one. This is a quantum discontinuity.
So why can you not get a tan from a wood-burning fireplace? Imagine a pendulum in the wind. Ordinarily a pendulum will swing in such a situation, even when there is not a high wind. Suppose, however, that the pendulum is allowed to absorb energy only in discrete steps of high denominations. In other words, it is a quantum pendulum. What then? Clearly, unless the wind is able to impart the required high increment of energy in one step, the pendulum will not move. Accepting energy in small denominations will not enable it to build up enough energy to cross a threshold. So it is with the jiggling electrons in a fireplace. Low-frequency radiation arises from small quantum jumps, but high frequency radiation requires large quantum jumps. A large quantum jump must be fueled by a large amount of energy in the electron’s environment; the energy in a wood-burning fireplace simply is not strong enough to create the conditions even for much blue light, let alone ultraviolet. That is the reason you cannot get a tan from a fireplace.
You may object. What happens when the ball falls from one step to another? Is it not taking on an intermediate position during the descent? This is where the strangeness of quantum theory enters: For a ball on a set of stairs, the answer is obviously yes, but for a quantum ball (an atom or an electron), Planck’s theory answers no. A quantum ball will never be found in any place intermediate between two steps; it is either on this one or on that one. This is a quantum discontinuity.
So why can you not get a tan from a wood-burning fireplace? Imagine a pendulum in the wind. Ordinarily a pendulum will swing in such a situation, even when there is not a high wind. Suppose, however, that the pendulum is allowed to absorb energy only in discrete steps of high denominations. In other words, it is a quantum pendulum. What then? Clearly, unless the wind is able to impart the required high increment of energy in one step, the pendulum will not move. Accepting energy in small denominations will not enable it to build up enough energy to cross a threshold. So it is with the jiggling electrons in a fireplace. Low-frequency radiation arises from small quantum jumps, but high frequency radiation requires large quantum jumps. A large quantum jump must be fueled by a large amount of energy in the electron’s environment; the energy in a wood-burning fireplace simply is not strong enough to create the conditions even for much blue light, let alone ultraviolet. That is the reason you cannot get a tan from a fireplace.

Introduction to Quantum Mechanics & NonDuality
Beginning from the End. (Radical Oneness/One Substance/Sunya/Consciousness, Timelessness, Always Already Is-ness, All things spontaneously Enlightened, Nothing Happens - Resolves all pairs of opposites, Complex mathematics Nondual, Geometry Nondual).
Introduction
Success of Theory - Theory behind all theories
Applications Abound - Computers, Cell phones, lasers, etc.
Taste of Quantum Weirdness
Best Theory Science Has for Ultimate Reality
Main Ingredients of QM are 2-fold
1) Evolution of Quantum System encoded by Schrodinger Equation/Wavefunctions
2) Measurement of the Quantum System by a Conscious Observer 'collapsing' it into one of its possible states.
Schrodinger Equation
Wave Particle Duality - Double Slit Experiment
I. Superposition States - Potential - No Objects= No Subjects = Nonduality
WaveFunction, Encodes, position, momentum, energy, etc.
1) Quantization - h - planck
2) Superposition ΣAi*Ѱ(x) - Eigenstates of Wavefunction
3) Uncertainty Principle
4) Orbitals of Atoms S, P, L, M Full expression of Wavefunctions
5) Identical Particles of Pauli Exclusion
Choice/Decision comes in Wholes
II. Collapse - Measurement - REALITY - Object Manifests = Subject Observing = Duality
0) Observer Effect - Consciousness creates reality
5) Probability P(i) = |Ai|^2 (not deterministic). Modifications of classical probability law.
III. Entanglement and Quantum Systems
7) Entanglement (connected to Entropy and information).
9) Quantum Coherence, Pure States, Mixed States and Entropy.
Beginning from the End. (Radical Oneness/One Substance/Sunya/Consciousness, Timelessness, Always Already Is-ness, All things spontaneously Enlightened, Nothing Happens - Resolves all pairs of opposites, Complex mathematics Nondual, Geometry Nondual).
Introduction
Success of Theory - Theory behind all theories
Applications Abound - Computers, Cell phones, lasers, etc.
Taste of Quantum Weirdness
Best Theory Science Has for Ultimate Reality
Main Ingredients of QM are 2-fold
1) Evolution of Quantum System encoded by Schrodinger Equation/Wavefunctions
2) Measurement of the Quantum System by a Conscious Observer 'collapsing' it into one of its possible states.
Schrodinger Equation
Wave Particle Duality - Double Slit Experiment
I. Superposition States - Potential - No Objects= No Subjects = Nonduality
WaveFunction, Encodes, position, momentum, energy, etc.
1) Quantization - h - planck
2) Superposition ΣAi*Ѱ(x) - Eigenstates of Wavefunction
3) Uncertainty Principle
4) Orbitals of Atoms S, P, L, M Full expression of Wavefunctions
5) Identical Particles of Pauli Exclusion
Choice/Decision comes in Wholes
II. Collapse - Measurement - REALITY - Object Manifests = Subject Observing = Duality
0) Observer Effect - Consciousness creates reality
5) Probability P(i) = |Ai|^2 (not deterministic). Modifications of classical probability law.
III. Entanglement and Quantum Systems
7) Entanglement (connected to Entropy and information).
9) Quantum Coherence, Pure States, Mixed States and Entropy.

The double slit experiment is absolutely impossible to explain in any classical way and has in it the heart of quantum mechanics.
In reality, it contains the only mystery."
- Richard Feynman
A lot can be learned conceptually about Quantum Mechanics from the double slit actual experiment and Schrondinger's Cat thought experiment. All the central mysteries of quantum mechanics lurk in these two examples, such as the wave-particle duality, the uncertainty principle, superposition, entanglement, and the measurement problem. If you deeply understand this experiment, even conceptually, you'll be on firm ground in understanding how quantum mechanics confirms the non-dual nature of the Universe
In reality, it contains the only mystery."
- Richard Feynman
A lot can be learned conceptually about Quantum Mechanics from the double slit actual experiment and Schrondinger's Cat thought experiment. All the central mysteries of quantum mechanics lurk in these two examples, such as the wave-particle duality, the uncertainty principle, superposition, entanglement, and the measurement problem. If you deeply understand this experiment, even conceptually, you'll be on firm ground in understanding how quantum mechanics confirms the non-dual nature of the Universe
 Gun spraying bullets towards a target
Gun spraying bullets towards a target
The Central Mystery of Quantum Mechanics
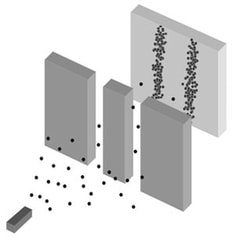
A gun (obeying classical physics) sprays bullets towards a target. Before they reach the target, they must pass through a screen with two slits. If bullets go through the slits they will most likely land directly behind the slit, but if they come in at a slight angle, they will land slightly to the sides. The resulting pattern is a map of the likelihood of a bullet landing at each point.
A gun (obeying classical physics) sprays bullets towards a target. Before they reach the target, they must pass through a screen with two slits. If bullets go through the slits they will most likely land directly behind the slit, but if they come in at a slight angle, they will land slightly to the sides. The resulting pattern is a map of the likelihood of a bullet landing at each point.
The above two-slit pattern happens to be simply the sum of the patterns for each slit considered separately: if half the bullets were fired with only the left slit open and then half were fired with just the right slit open, the result would be the same.
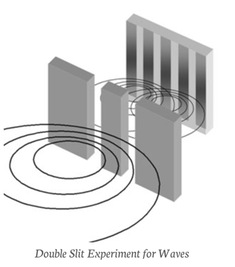
With waves, however, the result is very different, because of interference. If the slits were opened one at a time, the pattern would resemble that for bullets: two distinct peaks. But when both slits are open, the waves pass through both slits at once and interfere with each other: where they are in phase they reinforce each other; where they are out of phase they cancel each other out.
With waves, however, the result is very different, because of interference. If the slits were opened one at a time, the pattern would resemble that for bullets: two distinct peaks. But when both slits are open, the waves pass through both slits at once and interfere with each other: where they are in phase they reinforce each other; where they are out of phase they cancel each other out.
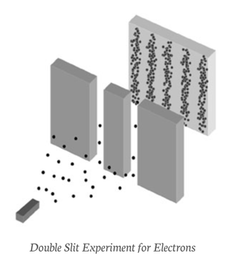
Now the quantum paradox:
Electrons, like bullets, strike the target one at a time. Yet, like waves, they create an interference pattern.
Electrons, like bullets, strike the target one at a time. Yet, like waves, they create an interference pattern.
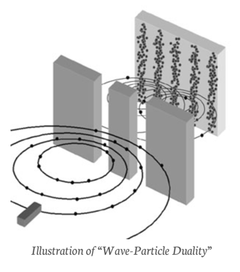
If each electron passes individually through one slit, with what does it “interfere?” Although each electron arrives at the target at a single place and time, it seems that each has passed through – or somehow felt the presence of both slits at once. Thus, the electron is understood in terms of a wave-particle duality.
The wave-particle duality is the central mystery of quantum mechanics–the one to which all others can ultimately be reduced.
The wave-particle duality is the central mystery of quantum mechanics–the one to which all others can ultimately be reduced.